Our groundbreaking work
-
Over 450,000
children cared for
Providing care for children
Each year, we provide care to more than 450,000 children and young people
-
90%
recommend Alder Hey
A great place to receive care
90% of staff would recommend Alder Hey as a place for friends and family to receive care
-
Top 20
ranking in the world
Top ranked paediatric hospital
Alder Hey is ranked in the top 20 of the best specialised paediatric hospitals in the world by Newsweek
-
£65m
raised for our charity
Making a difference since 2013
Since 2013, over £65m has been raised through our Charity, making a real difference to families every day
Latest news from Alder Hey
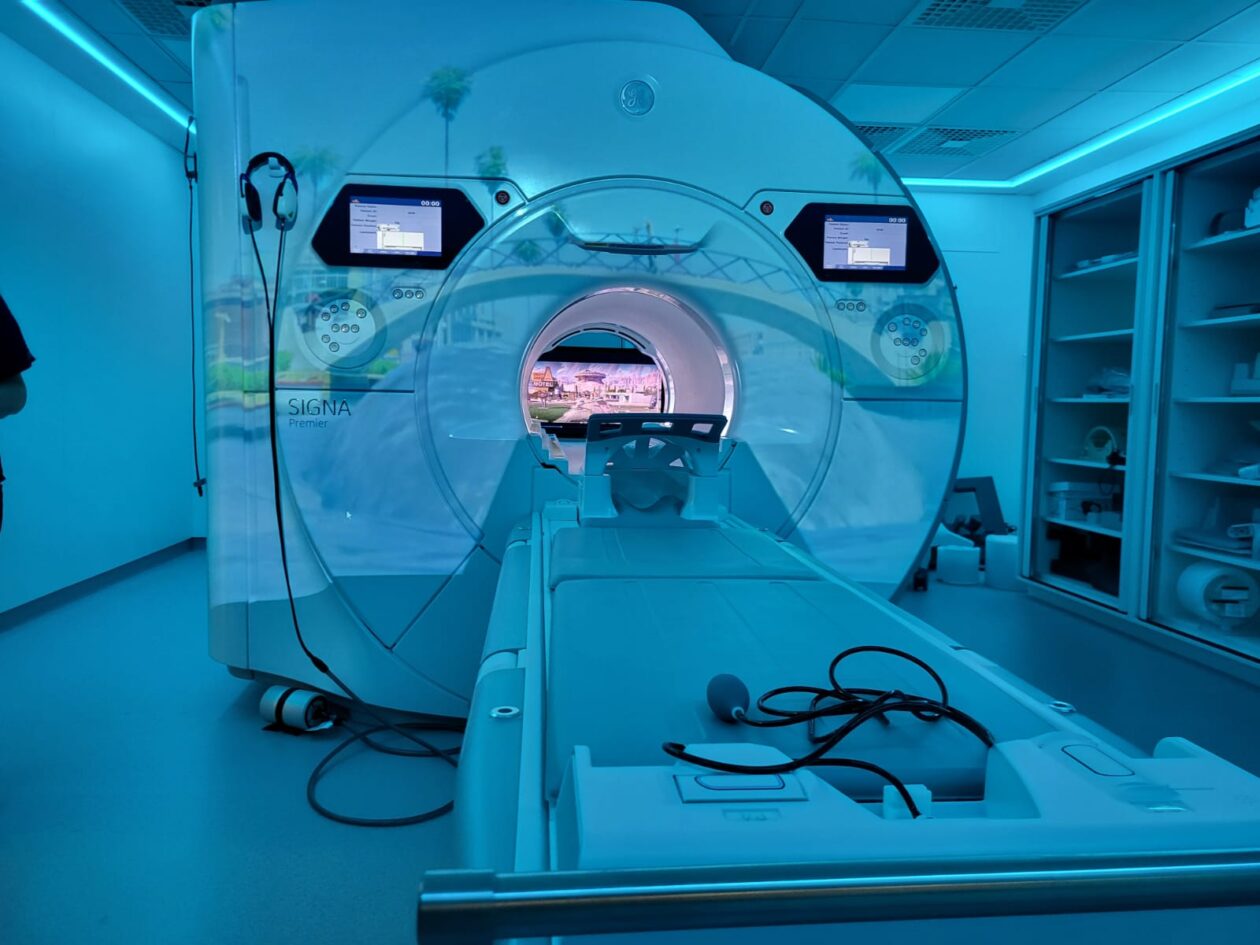
Book private paediatric MRI scans at Alder Hey
We offer a specialised private paediatric MRI service for children and young people aged 5–18.



